OUR SCIENCE
Platelets are key regulators of vascular integrity, bridging thrombosis, inflammation, and immunity. Advanced in vivo models and molecular tools are unlocking deeper insights and enable the development of safer, next-generation therapies.
Central players of hemostasis and thrombosis
Platelets are small, anucleate blood cells that continuously patrol the vascular system to maintain vessel integrity and enable blood clotting. They are short-lived and constantly replenished through thrombopoiesis, a process in which large, polyploid bone marrow–resident megakaryocytes release cytoplasmic fragments into the circulation.
Upon vascular injury, platelets encounter exposed extracellular matrix components, adhere to the damaged site, become activated, and—together with the coagulation cascade—form a fibrin-rich clot. This process, termed hemostasis, limits blood loss, prevents infection, and initiates tissue repair. When dysregulated, particularly in diseased vessels, this system can lead to intravascular clot formation (thrombosis), causing life-threatening events such as myocardial infarction or ischemic stroke. Cardiovascular diseases remain the leading cause of death worldwide, accounting for approximately 19 million deaths annually.[1]
Although antiplatelet and anticoagulant therapies are indispensable in preventing and treating thrombosis, all currently available agents carry an inherent risk of bleeding complications.[2]
- Martin SS, Aday AW, Almarzooq ZI et al.; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. 2024 Heart Disease and Stroke Statistics: A Report of US and Global Data From the American Heart Association. Circulation. 2024 149(8):e347-e913. doi: 10.1161/CIR.0000000000001209.
- van der Meijden PEJ, Heemskerk JWM. (2019) Platelet biology and functions: new concepts and clinical perspectives. Nat Rev Cardiol. 16(3):166-179. doi: 10.1038/s41569-018-0110-0.
Thrombo-inflammation
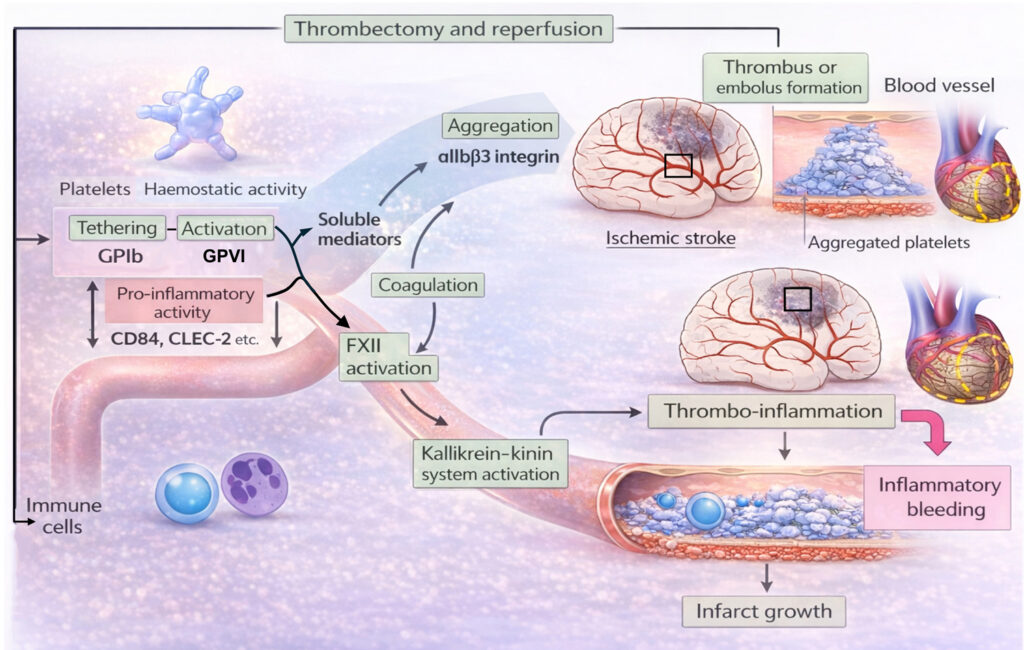
Importantly, platelets are also essential for maintaining vascular integrity in inflammatory settings—a process termed inflammatory hemostasis. This function operates partly independently of classical platelet activation pathways and involves ITAM-coupled receptors, integrin αIIbβ3, and additional organ-specific mechanisms that are incompletely understood.
- Nieswandt B, Kleinschnitz C, Stoll G. (2011) Ischaemic stroke: a thrombo-inflammatory disease? J Physiol. 589(17):4115-23. doi: 10.1113/jphysiol.2011.212886.
- Stoll G, Nieswandt B. (2019) Thrombo-inflammation in acute ischaemic stroke – implications for treatment. Nat Rev Neurol. 15(8):473-481. doi: 10.1038/s41582-019-0221-1.
As a key model in platelet research
Despite their biomedical importance, advances in understanding platelet function and the search for novel potential target proteins for platelet inhibition were long inefficient due to several limitations. First, the anucleate nature of platelets renders them inaccessible for genomic profiling and classic genetic methods for studies on protein function. Second, the myriad of haemodynamic and spatio-temporal cellular and molecular interactions that occur during thrombus formation and thrombo-inflammation in vivo cannot be mimicked in vitro. This changed dramatically around the turn of the century with i) the advent of genetic engineering technologies allowing the targeted manipulation of protein expression or function in murine megakaryocytes and platelets, and ii) the development of murine disease models and imaging approaches to study platelet/megakaryocyte function in the living organism.
Emfret Analytics has facilitated these developments by providing the scientific community with essential molecular tools allowing visualization, modulation, and quantitative analysis of platelet/megakaryocyte function in mice. This has led to fundamental discoveries that layed the ground for an entirely new understanding of the platelet/megakaryocyte system in health and disease and opened new avenues for the development of next-generation therapies tailored to target its pathological derailment while leaving physiological functions intact.
Platelet GPVI
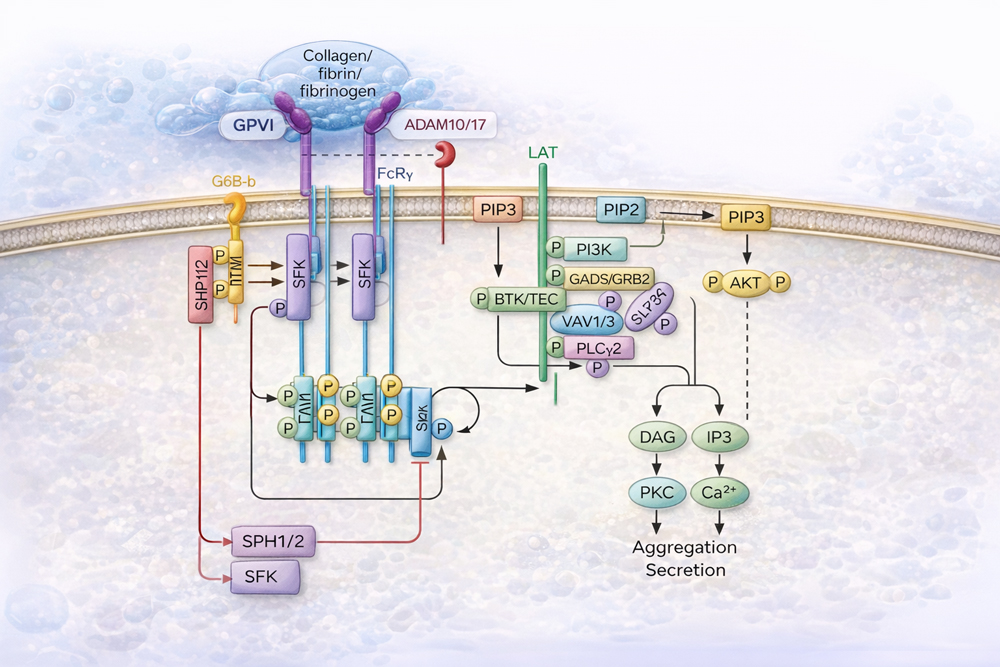
GPVI is a signal-transducing, multi-ligand immune-type receptor exclusively expressed on platelets (Figure). It orchestrates platelet activation and effector responses under pathological conditions while playing only a limited role in physiological hemostasis. This functional dissociation makes GPVI a highly attractive therapeutic target.
GPVI-mediated platelet activation contributes to organ damage across a broad spectrum of ischemic, inflammatory, autoimmune, and malignant diseases. In relevant preclinical models, Inhibition of GPVI has demonstrated robust protection without compromising normal hemostasis. EMA601, Emfret’s lead candidate, has shown potent antithrombotic and anti–thrombo-inflammatory efficacy in humanized preclinical models, supporting its potential as a next-generation therapeutic for acute vascular emergencies. [1,2]
- Navarro S, Talucci I, Göb V, Hartmann S, Beck S, Orth V, Stoll G, Maric HM, Stegner D, Nieswandt B (2024) The humanized platelet glycoprotein VI Fab inhibitor EMA601 protects from arterial thrombosis and ischaemic stroke in mice. Eur Heart J. 45(43):4582-97. doi: 10.1093/eurheartj/ehae482.
- McFadyen JD, Wang X, Peter K (2024). The quest for the holy grail in antithrombotic therapy: revitalized hope for platelet GPVI as a safe and effective antithrombotic target. Eur Heart J. 45(43):4598-4600. doi: 10.1093/eurheartj/ehae592.
Get in touch

-
EMFRET Analytics GmbH & Co. KG
Am Morgenroth 6
97246 Eibelstadt
Germany - service@emfret.com
